My research interests focus on protein folding and stability. Of particular interest to me are examples of protein folding that stretch the boundaries of what we know by presenting exceptions to the basic principles of protein folding learned in first semester biochemistry. Protein chaperones present such a topic. They "break" the rules by helping to determine both the structure and function of other proteins.
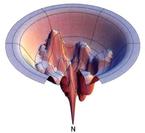
Figure from Dill &Chan (1997) Nat. Struct. Biol. Vol. 4, pp. 10-19
Figure from Dill &Chan (1997) Nat. Struct. Biol. Vol. 4, pp. 10-19
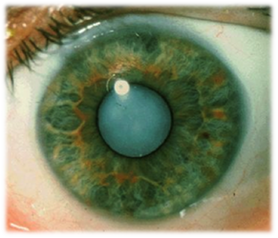
Alpha crystallin and cataracts
Over the past three years I have established a new project studying the oligomerization and protein chaperone function of Alpha Crystallin (aX) using spectroscopy and molecular biology approaches. The ability of aX to populate a range of oligomeric sizes in solution was the exceptional aspect of this system that drew me to it. Work on this project has been a team effort by many excellent undergraduate students at Amherst College and Austin College who have taken part in semester research, summer research projects, and Thesis work. This new project has resulted in five research posters and a presentation at the Annual Biophysical Society meetings from 2012-14 and a talk at the Protein Folders and Function meeting in 2015. Work investigating the mechanism of chaperon function is currently being refined and is being written up for publication in the coming months.
|
Images from: http://www.sightsurgeryinternational.com/cataracts_explained.html
|